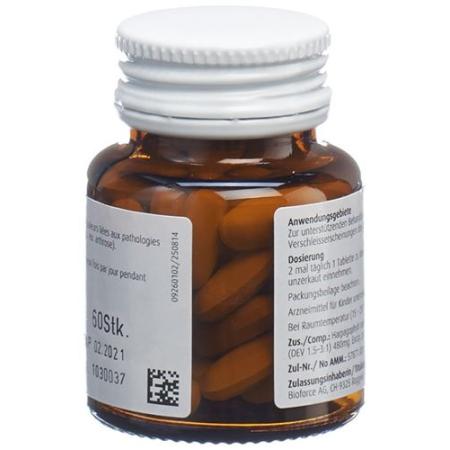
HARPAGOMED tablets
A. Vogel Harpagomed Rheumatabletten 60 Stück
Send a request to get a price
In stock 50 pcsAnalogues of the product based on the Anatomical Therapeutic Chemical (ATC) Classification - M01AX99
| Name | Code | EAN | Price | Original | ||
|---|---|---|---|---|---|---|
 |
HARPAGOMED tablets | 3474810 | 7680578710018 | - | Read more | |
 |
HARPAGOMED tablets | 3509721 | 7680578710032 | - | Read more | |
 |
Arkocaps Devil's Claw Caps vegetable 45 pcs | 3297176 | 7680570350014 | - | Read more | |
 |
Arkocaps Devil's Claw Caps vegetable 150 pcs | 3297182 | 7680570350021 | - | Read more | |
 |
Phytopharma devil's claw capsules 435 mg 45 pcs | 3642472 | 7680582720010 | - | Read more | |
 |
Phytopharma Teufelskralle Kapseln 435mg Pfl 150 Stück | 7836846 | 7640106953928 | - | Read more | |
 |
Dixa Teufelskrallenwurzel 250 tablets | 5454349 | 7680564250016 | - | Read more | |
 |
Vogel Arosan devil's claw tablets glass 120 pcs | 5582484 | 7680570510036 | - | Read more | |
 |
Vogel Arosan devil's claw tablets glass 60 pcs | 5598551 | 7680570510029 | - | Read more |
Description - HARPAGOMED tablets
HarpagoMed tablets contain devil's claw root extract (Harpagophytum procumbens D.C. and/or Harpagophytum zeyheri Decne). HarpagoMed tablets can be used to support the treatment of pain associated with degenerative diseases of the musculoskeletal system (e.g. arthrosis).
Swissmedic-approved patient information
HarpagoMed®
Herbal medicine
What are HarpagoMed tablets and when are they used?
HarpagoMed tablets contain extract of devil's claw root (Harpagophytum procumbens D.C. and/ or Harpagophytum zeyheri Decne). HarpagoMed tablets can be used to support the treatment of pain associated with degenerative diseases of the musculoskeletal system (e.g. arthrosis).
What should be considered?
A doctor should be consulted in the event of acute conditions, e.g . It can be used over a period of 2-3 months. After this time, please consult your doctor, pharmacist or druggist if the symptoms persist. This medicine contains lactose. Note for diabetics: 1 daily dosage (=2 film tablets) corresponds to 0.03 BE (0.03 BW).
When should HarpagoMed tablets not be used or should only be used with caution?
HarpagoMed tablets may be used in the event of hypersensitivity to devil's claw or any of the other ingredients of the drug, as well as gastric and duodenal ulcers not be taken. If you suffer from gallstones, you need to consult your doctor. There is no knowledge available on the use of the preparation in children. HarpagoMed rheumatism tablets should therefore not be used in children under the age of 12. Tell your doctor, pharmacist or druggist if you suffer from other illnesses, have allergies or are taking other medicines (including those you bought yourself).
Can HarpagoMed tablets be taken during pregnancy or breastfeeding?
There are no adequate studies on the use of this medicine during pregnancy and breastfeeding. It should therefore not be used during pregnancy and breastfeeding.
How do you use HarpagoMed tablets?
Adults: Take 1 tablet whole twice a day with meals with a little water. Experience has shown that it takes about 2-4 weeks until the full effectiveness can be expected. Follow the dosage given in the package leaflet or as prescribed by your doctor. If you think the medicine is too weak or too strong, talk to your doctor, pharmacist or druggist.
What side effects can HarpagoMed tablets have?
Slight gastrointestinal complaints (e.g. heartburn, flatulence, diarrhea, nausea with vomiting), headaches and allergic skin reactions can often occur . In the event of allergic reactions, treatment with HarpagoMed Rheumatism tablets should be discontinued and a doctor consulted. If you notice side effects that are not described here, you should inform your doctor, pharmacist or druggist.
What else needs to be considered?
HarpagoMed tablets should be stored at room temperature (15 ? 25° C) and out of the reach of children. HarpagoMed rheumatism tablets may only be used up to the date marked «EXP» on the container. Once opened, the pack should be used within 5 months. Unused or expired packs should be returned to your pharmacist or druggist. Your doctor, pharmacist or druggist can provide you with further information.
What does HarpagoMed tablets contain?
1 film-coated tablet contains: 480 mg dry extract from South African devil's claw root (DEV 1.5 - 3:1), extractant 60% V/V ethanol. This preparation also contains auxiliary substances.
Approval number
57871 (Swissmedic)
Where can you get HarpagoMed tablets? What packs are available?
In pharmacies and drugstores, without a doctor's prescription, in packs of 30, 60 and 120 tablets.
Authorization holder
A.Vogel AG, CH-9325 Roggwil This leaflet was last checked by the Medicines Agency (Swissmedic) in August 2015.
Description for the doctor
Pflanzliches Arzneimittel
Zusammensetzung
1 Filmtablette enthält: 480 mg Trockenextrakt aus der südafrikanischen Teufelskrallenwurzel (DEV 1.5–3:1), Auszugsmittel 60% V/V Ethanol. Dieses Präparat enthält zusätzlich Hilfsstoffe.
Dieses Arzneimittel enthält Lactose.
Hinweis für Diabetiker: 1 Tagesdosierung (= 2 Filmtabletten) entspricht 0,03 BE (0,03 BW).
Indikationen/Anwendungsmöglichkeiten
Unterstützende Behandlung von Schmerzen bei Verschleisserkrankungen des Bewegungsapparates (z.B. Arthrose).
Dosierung/Anwendung
Erwachsene: 2 mal täglich 1 Tablette zu den Mahlzeiten mit etwas Wasser unzerkaut einnehmen.
Die Anwendung kann über einen Zeitraum von 2–3 Monaten erfolgen.
Erfahrungsgemäss dauert es ca 2-4 Wochen bis die volle Wirksamkeit erwartet werden kann.
Kontraindikationen
Überempfindlichkeit gegen Teufelskralle oder einen der sonstigen Bestandteile des Arzneimittels, sowie Magen- und Zwölffingerdarmgeschwüre.
Zur Anwendung vom Präparat bei Kindern liegt kein Erkenntnismaterial vor. HarpagoMed Tabletten sollten deshalb bei Kindern unter 12 Jahren nicht angewendet werden.
Warnhinweise und Vorsichtsmassnahmen
Bei Gallensteinleiden ist Rücksprache mit dem Arzt erforderlich.
Schwangerschaft/Stillzeit
Zur Anwendung dieses Arzneimittels in Schwangerschaft und Stillzeit liegen keine ausreichenden Untersuchungen vor. Es sollte deshalb während der Schwangerschaft und Stillzeit nicht angewendet werden.
Unerwünschte Wirkungen
Häufig können leichte Magen-Darm-Beschwerden (z.B. Sodbrennen, Blähungen, Durchfall, Übelkeit mit Erbrechen), Kopfschmerzen sowie allergische Hautreaktionen auftreten.
Bei allergischen Reaktionen sollte die Behandlung mit HarpagoMed abgebrochen werden.
Sonstige Hinweise
HarpagoMed Tabletten sollen bei Raumtemperatur (15–25 °C) und ausserhalb der Reichweite von Kindern aufbewahrt werden.
HarpagoMed Rheuma-Tabletten dürfen nur bis zu dem auf dem Behälter mit «EXP» bezeichneten Datum verwendet werden.
Nach Anbruch soll die Packung innerhalb von 5 Monaten aufgebraucht werden.
Zulassungsnummer
57871 (Swissmedic).
Zulassungsinhaberin
A.Vogel AG, CH-9325, Roggwil
Stand der Information
August 2015.