Tobradex Susp Opht Drop Bottle 5ml
Tobradex 5 ml Augentropfen
Send a request to get a price
In stock 1000 pcsAnalogues of the product based on the Anatomical Therapeutic Chemical (ATC) Classification - S01CA01
| Name | Code | EAN | Price | Original | ||
|---|---|---|---|---|---|---|
 |
Maxitrol Eye Ointment Tb 3.5 g | 315293 | 7680315660101 | - | Read more | |
 |
Maxitrol Gtt Opht Fl 5ml | 315287 | 7680315670179 | - | Read more | |
 |
Spersadex comp. Gtt Opht Fl 5 ml | 1555361 | 7680305140255 | - | Read more | |
 |
Tobradex Eye Ointment Tb 3.5 g | 1611711 | 7680507670192 | - | Read more | |
 |
Dexagenta-POS Gtt Opht Fl 5ml | 4686827 | 7680595800013 | - | Read more | |
 |
DUCRESSA Gtt Opht | 7840304 | 7680681650010 | - | Read more |
Description - Tobradex Susp Opht Drop Bottle 5ml
What is TobraDex eye drop suspension and when is it used?
TobraDex eye drop suspension with the active ingredients tobramycin (antibiotic) and dexamethasone (glucocorticoid with anti-inflammatory effect) is intended for the treatment of certain eye infections with simultaneous bacterial eye infections.TobraDex eye drop suspension requires a more stringent prescription and may only be used on prescription from a doctor.
What precautions should be taken?
During the entire duration of treatment with TobraDex eye drops, your doctor must regularly check your eyes.The antibiotic (tobramycin) contained in TobraDex eye drop suspension is not effective against all microorganisms that cause infections in the eye. The use of an incorrectly chosen or improperly dosed antibiotic can cause complications. Therefore, never use the medicine on your own for the treatment of other diseases or for other people. In the event of new infections later, you must not use TobraDex eye drops without consulting your doctor again.
When must TobraDex eye drops suspension not be used?
TobraDex eye drops suspension must not be used in the following cases:
- in the case of a known or suspected hypersensitivity (allergy) to an ingredient of TobraDex eye dropsin glaucoma (green star)with eye infections that cannot be treated with an antibiotic, e.g. with certain viral diseases of the cornea and conjunctivain ocular tuberculosisfor ulcers or injuries to the cornea (even after uncomplicated removal of foreign bodies)for fungal diseases (mycoses) or untreated parasitic infections of the eye
When should you be careful when using TobraDex eye drops suspension?
- If an allergic reaction develops, such as itching of the eyes, swelling of the eyelid, reddening of the eyes or generalized reactions such as reddening of the skin, itching, formation of wheals on the skin, anaphylaxis and blistering, discontinue therapy and consult your doctor Doctor. These allergic reactions can also occur with other treatments (topical or systemic) with an antibiotic of the same type (aminoglycosides). If you use other antibiotics together with TobraDex eye drops, ask your doctor.If you have or have ever had a condition such as myasthenia gravis or Parkinson's, ask your doctor for advice. These types of antibiotics can make muscle weakness worse.If TobraDex eye drops suspension is used for a long time, there is a risk of fungal infection.For purulent eye infections.Corticosteroids can mask, activate, or worsen an infection in the eye. If TobraDex eye drops suspension is used for a long time, this can lead to delayed wound healing. It can also lead to an increase in intraocular pressure and, as a result, to glaucoma. During the treatment with TobraDex eye drop suspension you should be under constant medical supervision, whereby the intraocular pressure should be measured regularly by the doctor.If the eye complaints treated with TobraDex eye drop suspension worsen after 2-3 days of treatment or if new symptoms occur, you should consult your doctor immediately.If you suffer from diseases which result in thinning of the tissues around the eye (cornea or sclera): Topical use of corticosteroids can cause perforation of the eye.If you have diabetes (especially type I diabetes) there is a risk of cataracts. The use of corticosteroids in diabetics increases the risk of early and rapidly occurring cataracts.If swelling occurs on the trunk and face (Cushing's syndrome), you should inform your doctor. This risk is particularly important in children and patients treated with ritonavir or cobicistat.TobraDex eye drop suspension contains benzalkonium chloride, a preservative that can cause eye irritation and discolour soft contact lenses. Avoid contact with soft lenses. During treatment with TobraDex eye drops, you must wait at least 15 minutes before the lenses can be reinserted.Benzalkonium chloride can also cause eye irritation, especially if you have dry eyes or diseases of the cornea (clear layer on the front of the eye). Contact your doctor if you get an unusual burning sensation, or pain in your eye after using this medicineIf TobraDex eye drop suspension and pupil-dilating drug are used at the same time, caution is advised, as an increase in intraocular pressure cannot be ruled out under these circumstances.The use and safety of TobraDex eye drops suspension in children and adolescents has not yet been tested. Therefore, the use of TobraDex eye drops suspension in children and adolescents is not recommended.When treating with more than one eye preparation, wait at least 5 minutes between each application. Eye ointments are to be used at the end.Inform your doctor or pharmacist if you suffer from other illnesses, have allergies or take other medicines (including those you have bought yourself) or use on your eyes.As blurred vision may occur immediately after using TobraDex eye drops, you should not drive or use machines until the symptoms have subsided.
Can TobraDex eye drops suspension be taken during pregnancy or while breastfeeding?
TobraDex eye drops suspension is not recommended during pregnancy.TobraDex eye drops, suspension is not recommended during breastfeeding, therefore you should either stop breastfeeding or discontinue the medicine.
How do you use TobraDex eye drop suspension?Dosage / applicationAdult
The dosage is determined individually by the ophthalmologist for each patient. In any case, adhere to the dosage determined for you by your doctor. Neither the daily dose nor the duration of therapy may be changed without consulting your doctor. In addition, the treatment must not be interrupted without instructions from your doctor. If swelling of the face or trunk (Cushing's syndrome) and / or extreme weakness, nausea and persistent diarrhea (symptoms of adrenal suppression) occur, the treatment should not be reduced abruptly but gradually, in consultation with your doctor, until the drug is discontinued .If too much TobraDex eye drop suspension has gotten into the eye, rinse the eye out with lukewarm water. Do not add any more drops until it is time for the next scheduled application.If you think that the medicine is too weak or too strong, talk to your doctor or pharmacist.The doctor can prescribe TobraDex eye ointment as an additional treatment to TobraDex eye drop suspension (TobraDex eye ointment is preferably used as a night treatment just before going to bed).
Children and adolescents
The use and safety of TobraDex eye drops suspension in children and adolescents has not yet been tested.
What side effects can TobraDex eye drops suspension have?
The following side effects can occur when using TobraDex eye drops:
Uncommon (affects 1 to 10 users in 1,000)
Increase in intraocular pressure, eye pain, eye irritation, itching of the eye, discomfort of the eye.
Rare (affects 1 to 10 in 10,000 users)
Inflammation of the cornea, allergic reactions (e.g. itching or swelling of the eyelids), blurred vision, dry eyes, reddening of the eyes, taste disturbances.If such symptoms occur, your ophthalmologist should be consulted immediately.
Post-marketing experience shows the following side effects
Eyelid swelling or redness, dilated pupils, increased tearing, hypersensitivity, dizziness, headache, nausea, abdominal discomfort and rash, swollen face, itching, skin reactions (erythema multiforme).
Further side effects of the individual active ingredients that can potentially occur with TobraDex eye dropsTobramycin
Corneal abrasion, decreased visual acuity, conjunctival swelling, eye discharge, skin inflammation, loss of eyelashes, skin whitening, dry skin.
Dexamethasone
Conjunctivitis, visible defects due to the discoloration of the cornea, sensitivity to light, foreign body sensation, abnormal sensation in the eye, crusting of the eyelid margin, irritation, glaucoma, ulcer-causing corneal inflammation, reduced visual acuity, corneal erosion, drooping of the eyelid. Hormonal changes, e.g. increased body hair (especially in women), muscle weakness and atrophy, stretch marks on the skin, high blood pressure, irregular or absent menstruation, changes in protein and calcium concentration in the body, short stature in children and teenagers as well as swelling and increase in the torso and Face (Cushing syndrome).If any side effects occur which you suspect to be related to the use of TobraDex eye drops, you should inform your doctor or pharmacist immediately.If you notice any side effects that are not described here, you should inform your doctor or pharmacist.
What should also be noted?
The medicinal product may only be used up to the expiry date marked ?Use by? on the container.After the end of the treatment, please bring the remainder of the medicinal product to your dispensing point (doctor or pharmacist) for proper disposal.Your doctor or pharmacist can provide you with further information. These people have the detailed information for specialists.
Use-by period after opening
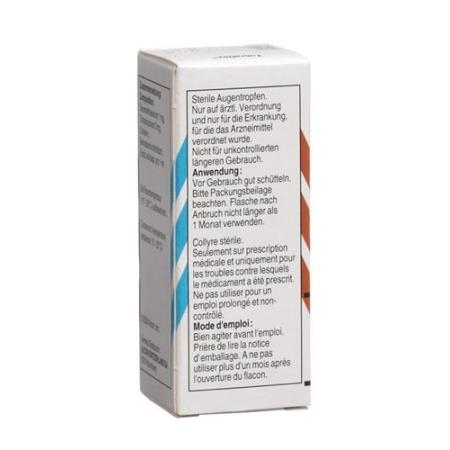
The contents must not be used for more than 30 days after opening the bottle.
Storage advice
Store the bottle upright in the original packaging at room temperature (15-25 ° C) out of the reach of children.
More information
Shake bottle well before use. To avoid possible microbial contamination of the eye drops, the tip of the bottle must not come into contact with your hands, eyes or any other object. Close the bottle immediately after use and always keep it tightly closed.After opening the bottle, remove the loose safety ring before using the drops.
What does TobraDex eye drop suspension contain?Active ingredients
1 ml eye drop suspension contains dexamethasone 1.0 mg, tobramycin 3.0 mg
Auxiliary materials
This medicinal product contains 0.1 mg benzalkonium chloride per 1 ml solution. Other excipients are tyloxapol, disodium edetate, sodium chloride, hydroxyethyl cellulose, sodium sulfate, sulfuric acid and / or sodium hydroxide, purified water.
Approval number
50766 (Swissmedic)
Where can you get TobraDex eye drop suspension? Which packs are available?
Available in pharmacies against a doctor's prescription, which only entitles the holder to purchase once.
- Tobradex eye drops 5 ml
Marketing authorization holder
Novartis Pharma Schweiz AG, Risch; Domicile: 6343 Rotkreuz.